This article first appeared in The Checkup, MIT Technology Review’s weekly biotech newsletter. To receive it in your inbox every Thursday, and read articles like this first, sign up here.
Therapies to treat brain diseases share a common problem: they struggle to reach their target. The blood vessels that permeate the brain have a special lining so tightly packed with cells that only very tiny molecules can pass through. This blood-brain barrier “acts as a seal,” protecting the brain from toxins or other harmful substances, says Anne Eichmann, a molecular biologist at Yale. But it also keeps most medicines out. Researchers have been working on methods to sneak drugs past the blood-brain barrier for decades. And their hard work is finally beginning to pay off.
Last week, researchers at the West Virginia University Rockefeller Neuroscience Institute reported that by using focused ultrasound to open the blood-brain barrier, they
improved delivery of a new Alzheimer’s treatment and sped up clearance of the sticky plaques that are thought to contribute to some of the cognitive and memory problems in people with Alzheimer’s by 32%.
For this issue of The Checkup, we’ll explore some of the ways scientists are trying to disrupt the blood-brain barrier.

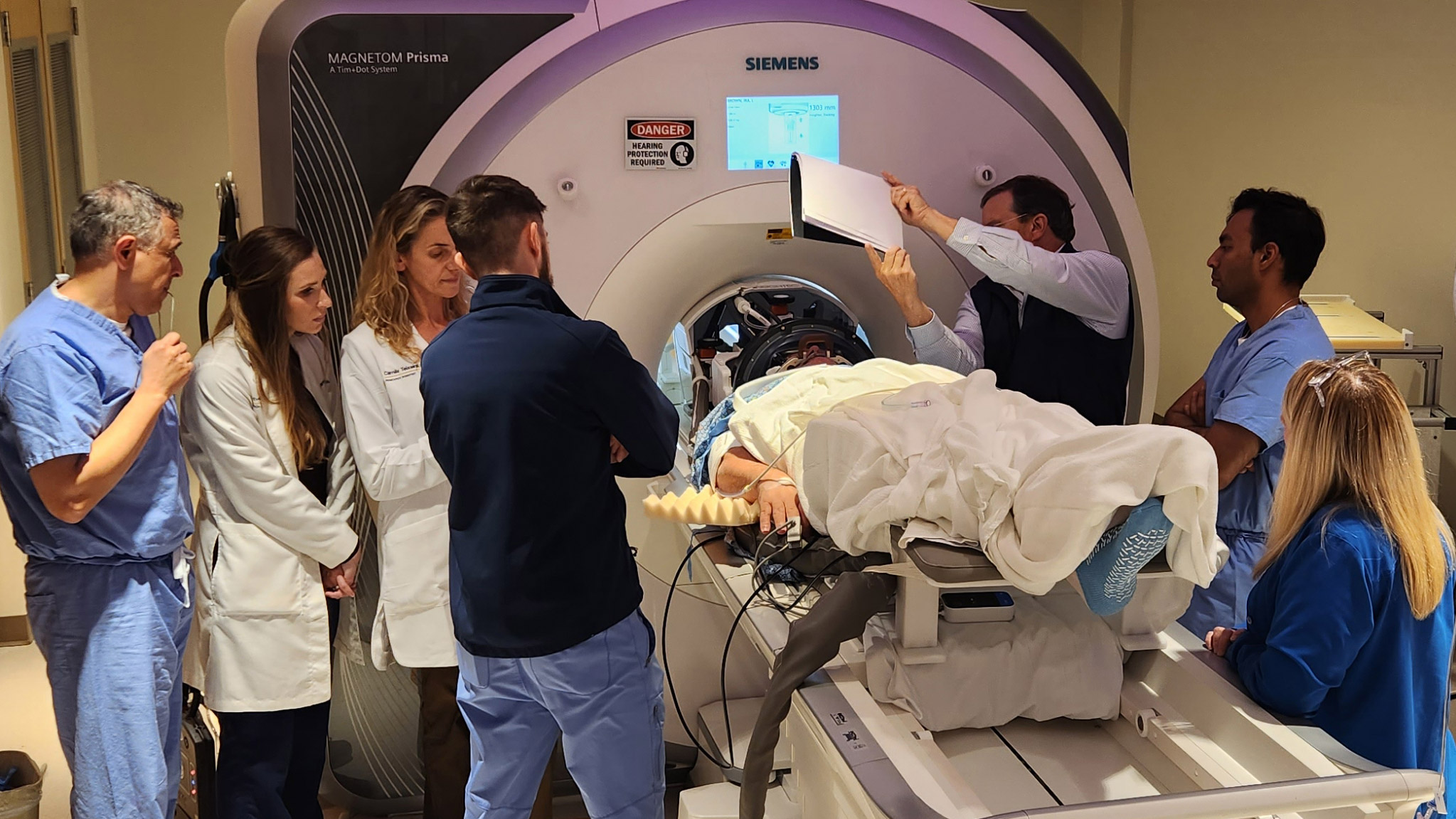
In the West Virginia study, three people with mild Alzheimer’s received monthly doses of aducanumab, a lab-made antibody that is delivered via IV. This drug, first approved in 2021, helps clear away beta-amyloid, a protein fragment that clumps up in the brains of people with Alzheimer’s disease. (The drug’s approval was controversial, and it’s still not clear whether it actually slows progression of the disease.) After the infusion, the researchers treated specific regions of the patients’ brains with focused ultrasound, but just on one side. That allowed them to use the other half of the brain as a control. PET scans revealed a greater reduction in amyloid plaques in the ultrasound-treated regions than in those same regions on the untreated side of the brain, suggesting that more of the antibody was getting into the brain on the treated side.
Aducanumab does clear plaques without ultrasound, but it takes a long time, perhaps in part because the antibody has trouble entering the brain. “Instead of using the therapy intravenously for 18 to 24 months to see the plaque reduction, we want to see if we can achieve that reduction in a few months,” says Ali Rezai, a neurosurgeon at West Virginia University Rockefeller Neuroscience Institute and an author of the new study. Cutting the amount of time needed to clear these plaques might help slow the memory loss and cognitive problems that define the disease.
The device used to target and deliver the ultrasound waves, developed by a company called Insightec, consists of an MRI machine and a helmet studded with ultrasound transducers. It’s FDA approved, but for an entirely different purpose: to help stop tremors in people with Parkinson’s by creating lesions in the brain. To open the blood-brain barrier, “we inject individuals intravenously with microbubbles,” Rezai says. These tiny gas bubbles, commonly used as a contrast agent, travel through the bloodstream. Using the MRI, the researchers can aim the ultrasound waves at very specific parts of the brain “with millimeter precision,” Rezai says. When the waves hit the microbubbles, the bubbles begin to expand and contract, physically pushing apart the tightly packed cells that line the brain’s capillaries. “This temporary opening can last up to 48 hours, which means that during those 48 hours, you can have increased penetration into the brain of therapeutics,” he says.
Focused ultrasound has been explored as a method for opening the blood-brain barrier for years. (We wrote about this technology way back in 2006.) But this is the first time it has been combined with an Alzheimer’s therapy and tested in humans.
The proof-of-concept study was too small to look at efficacy, but Rezai and his team are planning to continue their work. The next step is to repeat the study in five people with one of the newer anti-amyloid antibodies, lecanemab. Not only does that drug clear plaque, but one study showed that it slowed disease progression by about 30% after 18 months of treatment in patients with early Alzheimer’s symptoms. That’s a modest amount, but a major success in a field that has struggled with repeated failures.
Eichmann, who is also working on disrupting the blood-brain barrier, says the new results using focused ultrasound are exciting. But she wonders about long-term effects of the technique. “I guess it remains to be seen whether over time, upon repeated use, this would be damaging to the blood-brain barrier,” she says.
Other strategies for opening the blood-brain barrier look promising too. Rather than mechanically pushing the barrier apart, Roche, a pharmaceutical company, has developed a technology called “Brainshuttle” that ferries drugs across it by binding to receptors on the cells that line the vessel walls.
The company has linked Brainshuttle to its own anti-amyloid antibody, gantenerumab, and is testing it in 44 people with Alzheimer’s. At a conference in October, researchers presented initial results. The highest dose completely wiped out plaque in three of four participants. The biotech company Denali Therapeutics is working on a similar strategy to tackle Parkinson’s and other neurodegenerative diseases..
Eichmann is working on a different strategy. Her team is testing an antibody that binds to a receptor that is important for maintaining the integrity of the blood-brain barrier. By blocking that receptor, they can temporarily loosen the junctions between cells, at least in lab mice.
Other groups are targeting different receptors, exploring various viral vectors, or developing nanoparticles that can slip into the brain.
All these strategies will have different advantages and drawbacks, and it isn’t yet clear which will be safest and most effective. But Eichmann thinks some strategy is likely to be approved in the coming years: “We are indeed getting close.”
Techniques to open the blood-brain barrier could be useful in a whole host of diseases—Alzheimer’s, but also Parkinson’s disease, ALS, and brain tumors. “This really opens up a whole array of potential opportunities,” Rezai says. “It’s an exciting time.”
Read more from MIT Technology Review’s archive
Until recently, drug development in Alzheimer’s had been a dismal pursuit, marked by repeated failures. In 2017, Emily Mullin looked at how failures of some of the anti-amyloid drugs had researchers questioning whether amyloid is really the problem in Alzheimer’s.
In 2016, Ryan Cross covered one of the first efforts to use ultrasound to open the blood-brain barrier in humans, a trial to deliver chemotherapy to patients with recurrent brain tumors. That same year, Antonio Regalado reported some of the first exciting results of the Alzheimer’s drug aducanumab.
From around the web
Bayer’s non-hormonal drug to treat hot flashes reduced their frequency and intensity and improved sleep and quality of life. These results, coupled with other recent advances in treatment for symptoms of menopause, are a sign that these long-neglected issues have become big business. (Stat)
Covid is surging. Wastewater data is the best way we have to measure the virus’s ebb and flow, but it’s far from perfect. (NYT)
Last week the FDA approved Florida’s request to import drugs from Canada to cut costs. The pharmaceutical industry is not thrilled. (Reuters) Neither is Canada. (Ars Technica)
Comments
Post a Comment